The current results indicate that PTG-300 is safe and well tolerated. It is an effective agent for
eliminating the need for TP in PV patients with low and high risk disease. Observation of no
phlebotomy for 7 months in the previously TP dependent patients is quite significant. The
effect of PTG 300 on PV-related symptoms is also being evaluated. Continued patient
enrollment and results of blinded withdrawal phase will provide more definitive conclusions
regarding the efficacy of PTG 300.
References:
1. Marchioli et al., Cardiovascular events and intensity of treatment in polycythemia vera. N Negl J Med
2013 Jan 3;368(1):22-33
2. Ginzburg YZ et al. Dysregulated iron metabolism in polycythemia vera: etiology and consequences
Leukemia. 2018 Oct;32(10):2105-2116
3. Casu et al., Minihepcidin peptides as disease modifiers in mice affected by β-thalassemia and
polycythemia vera. Blood. 2016 Jul 14;128(2):265-76
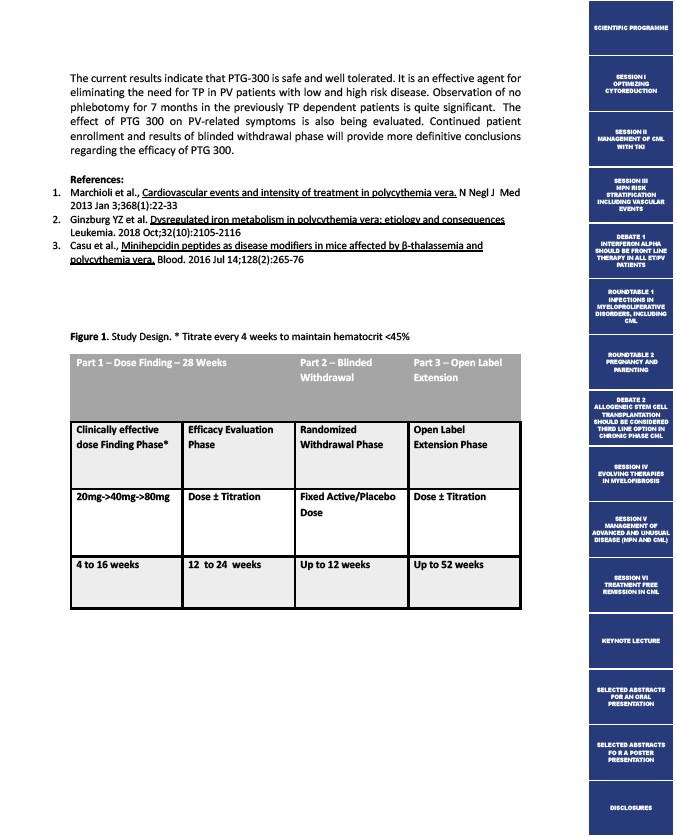
Figure 1. Study Design. * Titrate every 4 weeks to maintain hematocrit <45%
Part 1 ─ Dose Finding – 28 Weeks
Part 2 ─ Blinded
Withdrawal
Part 3 ─ Open Label
Extension
Clinically effective
dose Finding Phase*
Efficacy Evaluation
Phase
Randomized
Withdrawal Phase
Open Label
Extension Phase
20mg->40mg->80mg Dose ± Titration
Fixed Active/Placebo
Dose
Dose ± Titration
4 to 16 weeks
12 to 24 weeks
Up to 12 weeks
Up to 52 weeks
SCIENTIFIC PROGRAMME
SESSION I
OPTIMIZING
CYTOREDUCTION
SESSION II
MANAGEMENT OF CML
WITH TKI
SESSION III
MPN RISK
STRATIFICATION
INCLUDING VASCULAR
EVENTS
DEBATE 1
INTERFERON ALPHA
SHOULD BE FRONT LINE
THERAPY IN ALL ET/PV
PATIENTS
ROUNDTABLE 1
INFECTIONS IN
MYELOPROLIFERATIVE
DISORDERS, INCLUDING
CML
ROUNDTABLE 2
PREGNANCY AND
PARENTING
DEBATE 2
ALLOGENEIC STEM CELL
TRANSPLANTATION
SHOULD BE CONSIDERED
THIRD LINE OPTION IN
CHRONIC PHASE CML
SESSION IV
EVOLVING THERAPIES
IN MYELOFIBROSIS
SESSION V
MANAGEMENT OF
ADVANCED AND UNUSUAL
DISEASE (MPN AND CML)
SESSION VI
TREATMENT FREE
REMISSION IN CML
KEYNOTE LECTURE
SELECTED ABSTRACTS
FOR AN ORAL
PRESENTATION
SELECTED ABSTRACTS
FO R A POSTER
PRESENTATION
DISCLOSURES